Have you ever wondered whether NaCl (sodium chloride) — the salt we use every day — is ionic or covalent? Many students and even adults get confused by this chemistry question.
At first glance, NaCl might seem simple, but to understand whether it’s ionic or covalent, you need to know what these two bond types actually mean.
In this easy-to-understand guide, we’ll explain:
- What ionic and covalent bonds are,
- Why NaCl is ionic, not covalent,
- The difference between ionic and covalent bonds,
- Common examples and memory tricks, and
- A short quiz to test your understanding.
By the end, you’ll clearly know why table salt is an ionic compound and how to tell the difference in any other chemical formula too.
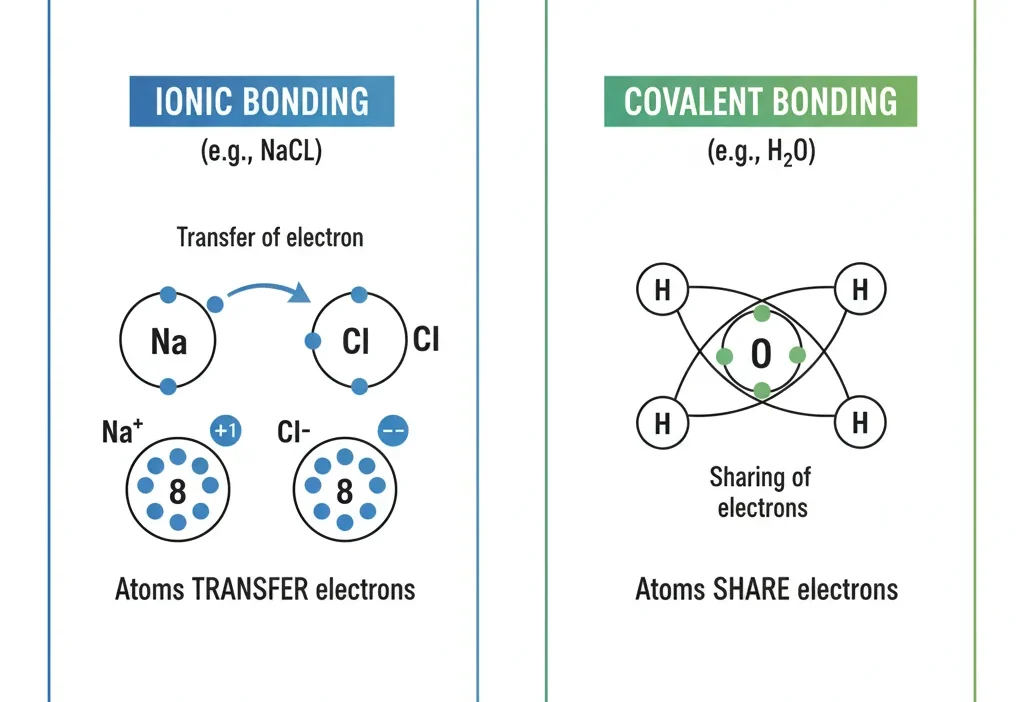
⚗️ What Does “Ionic” and “Covalent” Mean?
Let’s start with the basics. A chemical bond is what holds atoms together to make molecules or compounds. But not all bonds are the same — some share electrons, while others transfer them.
🔹 Ionic Bond – Simple Definition
An ionic bond happens when one atom gives away an electron to another atom.
- One atom becomes positive (cation).
- The other becomes negative (anion).
- Opposite charges attract, forming a strong ionic bond.
Example of Ionic Bonds:
- NaCl (Sodium Chloride) – Salt
- KBr (Potassium Bromide)
- MgO (Magnesium Oxide)
💡 Simple way to remember:
“Ionic bonds involve giving and taking.”
🔸 Covalent Bond – Simple Definition
A covalent bond happens when atoms share electrons equally.
- No giving or taking, just sharing.
- Usually occurs between nonmetals.
- They form neutral molecules (not charged).
Example of Covalent Bonds:
- H₂O (Water)
- CO₂ (Carbon Dioxide)
- O₂ (Oxygen Gas)
💡 Simple way to remember:
“Covalent bonds involve sharing.”
⚖️ The Key Difference Between Ionic and Covalent Bonds
Let’s compare them side-by-side for better understanding.
| Feature | Ionic Bond | Covalent Bond |
|---|---|---|
| How formed | Electrons are transferred | Electrons are shared |
| Between | Metal + Nonmetal | Nonmetal + Nonmetal |
| Example | NaCl, MgO | H₂O, CO₂ |
| Type of force | Strong electrostatic attraction | Sharing of electron pairs |
| Conducts electricity? | Yes (in water) | No |
| Physical form | Crystalline solids | Gases or liquids (mostly) |
| Melting point | High | Low to medium |
| Bond strength | Very strong | Moderate |
💡 Quick Tip to Remember:
If the compound has a metal and a nonmetal, it’s usually ionic.
If it has two nonmetals, it’s covalent.
🧂 Why NaCl Is Ionic, Not Covalent
Now that you know the basics, let’s look at NaCl — sodium chloride — closely.
- Sodium (Na) is a metal.
- Chlorine (Cl) is a nonmetal.
- When sodium meets chlorine, sodium gives one electron to chlorine.
- Sodium becomes Na⁺, and chlorine becomes Cl⁻.
- These opposite ions attract each other, forming a strong ionic bond.
That’s why NaCl is an ionic compound — the bond is based on electron transfer, not sharing.
⚡ Example Reaction
Na (metal) → Na⁺ + e⁻
Cl + e⁻ → Cl⁻
→ Na⁺ + Cl⁻ → NaCl
💡 Memory Hack:
“NaCl = Giving Salt” → Sodium gives its electron → Ionic bond.
❌ Common Mistakes and How to Avoid Them
Mistake 1: Thinking NaCl is Covalent Because It’s Neutral
NaCl may be neutral overall, but inside, it’s made of charged ions (Na⁺ and Cl⁻). The neutrality comes from the balance of charges, not shared electrons.
✅ Correct understanding: Neutrality doesn’t mean covalent — it just means balanced charges.
Mistake 2: Believing All Solid Compounds Are Ionic
Not true! Some covalent compounds (like sugar or wax) are also solid.
The key is not the state, but whether electrons are shared or transferred.
✅ Tip: Always check if a metal and nonmetal are involved — if yes, it’s ionic.
Mistake 3: Confusing NaCl with Cl₂
NaCl is ionic (sodium + chlorine).
Cl₂ is covalent (chlorine + chlorine).
The difference is in who bonds with whom.
✅ Tip:
Same elements bond covalently; different ones (metal + nonmetal) often bond ionically.
📘 When to Identify a Compound as Ionic
You should classify a bond as ionic when:
- It forms between a metal and a nonmetal.
- One atom loses electrons and another gains them.
- The compound forms ions (charged particles).
- It dissolves easily in water and conducts electricity.
Examples of Ionic Compounds:
- NaCl – Table salt
- KBr – Used in lab reagents
- CaCl₂ – Found in road salt
- MgO – Used in medicine
- LiF – Used in batteries
💡 Quick Clue:
If it forms crystals or conducts electricity in water, it’s likely ionic.
🔬 When to Identify a Compound as Covalent
A bond is covalent when:
- It forms between two nonmetals.
- Electrons are shared, not given away.
- It forms neutral molecules (no ions).
- The substance usually doesn’t conduct electricity.
Examples of Covalent Compounds:
- H₂O (Water) – Shared electrons between hydrogen and oxygen
- CO₂ (Carbon Dioxide) – Shared bonds between carbon and oxygen
- O₂ (Oxygen gas) – Shared electrons between two oxygen atoms
- CH₄ (Methane) – Shared bonds between carbon and hydrogen
- NH₃ (Ammonia) – Shared electrons between nitrogen and hydrogen
💡 Memory Trick:
Covalent = “Co-operate.” Atoms co-operate and share electrons.
🔁 Quick Recap: Ionic vs Covalent Bonds
Here’s a short summary you can keep in mind:
- Ionic bonds: Transfer electrons → metal + nonmetal → forms ions → strong attraction.
- Covalent bonds: Share electrons → nonmetal + nonmetal → forms molecules → moderate bond.
🧠 Easy Mnemonics
- “I Give = Ionic” (electrons are given away)
- “We Share = Covalent” (electrons are shared)
🧑🔬 Advanced Tips for Curious Learners
- Origin of the Terms:
- Ionic comes from “ion,” meaning a charged atom.
- Covalent comes from “co-” (together) + “valent” (strength), meaning sharing strength.
- In Exams:
- Always look at the periodic table:
- Metals on the left → form ionic bonds.
- Nonmetals on the right → form covalent bonds.
- Always look at the periodic table:
- In Everyday Life:
- Salt (NaCl) → ionic
- Sugar (C₆H₁₂O₆) → covalent
- Baking soda (NaHCO₃) → mostly ionic
- Water (H₂O) → covalent
- In Texting or Online Quizzes:
- “NaCl ionic or covalent” is a top-searched question in chemistry basics.
- The short answer is always: NaCl is ionic.
🧩 Mini Quiz: Test Yourself!
Fill in the blanks with ionic or covalent.
- NaCl is an ______ bond.
- H₂O is a ______ compound.
- In an ______ bond, electrons are transferred.
- In a ______ bond, electrons are shared.
- CO₂ has ______ bonds.
- KBr is an example of an ______ compound.
- Two oxygen atoms form a ______ bond.
✅ Answers: 1-Ionic, 2-Covalent, 3-Ionic, 4-Covalent, 5-Covalent, 6-Ionic, 7-Covalent.
❓ FAQs
1. Is NaCl ionic or covalent?
NaCl is ionic because sodium transfers an electron to chlorine, forming charged ions (Na⁺ and Cl⁻).
2. Why is NaCl not covalent?
Because the electrons aren’t shared — they are transferred. Covalent bonds involve sharing.
3. How can you tell if a bond is ionic?
If it’s between a metal and nonmetal, and electrons move from one atom to another, it’s ionic.
4. Is NaCl a molecule or a compound?
NaCl is a compound, not a molecule, because it’s held together by ionic bonds.
5. What’s an easy way to remember ionic vs covalent?
Ionic = “I give.”
Covalent = “We share.”
🧭 Conclusion
Understanding whether NaCl is ionic or covalent becomes easy once you know how atoms behave. Sodium gives its electron to chlorine — that’s a perfect example of ionic bonding.
Remember the golden rule:
Metal + Nonmetal → Ionic
Nonmetal + Nonmetal → Covalent
So next time you sprinkle salt on your food, you’ll know that each grain is made of billions of tiny ionic bonds!

Marianne Solace is a lifestyle and personal-growth writer for WordContrast.com. Her work blends inspiration with practicality, offering thoughtful insights on wellness, creativity, and mindful living. When she’s not writing, Marianne enjoys journaling with a cup of coffee, exploring art museums, and helping others find balance through the written word.