Many students and even adults often ask the same question: Is salt water homogeneous or heterogeneo? The words “homogeneous” and “heterogeneous” sound long and confusing, but their meanings are actually very simple.
People mix up “homogeneous” and “heterogeneous” because both are used to describe mixtures — things made by combining two or more substances. Salt water is one of the most common mixtures we see in daily life, so it’s the perfect example to understand the difference.
In this easy guide, you’ll learn:
- what “homogeneous” and “heterogeneous” mean
- which one salt water belongs to
- simple examples you can remember
- how to understand and use these words correctly in science or school
This explanation is so clear that even a fourth-grade student can understand it.
What Does “Homogeneous” Mean?
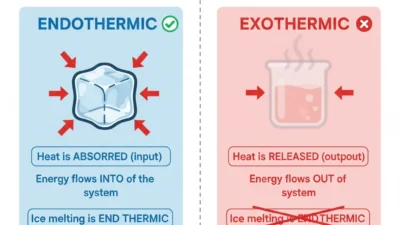
A homogeneous mixture is a mixture where everything is evenly mixed. You cannot see the different parts because they blend together completely.
Simple Meaning:
A homogeneous mixture looks the same everywhere.
Part of Speech:
Adjective (describes a mixture)
Examples of Homogeneous Mixtures:
- Salt water – The salt dissolves completely, so everything looks uniform.
- Sugar water – You cannot see sugar crystals.
- Air – Even though it’s made of many gases, it looks the same everywhere.
Mini-Story to Remember:
Imagine adding a spoon of salt to a glass of warm water. You stir and stir until you cannot see the salt anymore. No matter where you taste from—top or bottom—it tastes the same.
👉 This is homogeneous.
What Does “Heterogeneous” Mean?
A heterogeneous mixture is a mixture where the parts do not mix evenly. You can see the different substances.
Simple Meaning:
The mixture looks different in different parts.
Part of Speech:
Adjective (describes a mixture)
Examples of Heterogeneous Mixtures:
- Sand in water – Sand settles at the bottom.
- Fruit salad – You see pieces of apple, banana, grapes, etc.
- Cereal in milk – One part is crunchy, one part is soggy.
Mini-Story to Remember:
Think of a jar filled with stones and water. You can clearly see the stones.
👉 This is heterogeneous.
The Key Difference Between Homogeneous and Heterogeneous (Table)
| Feature | Homogeneous | Heterogeneous |
|---|---|---|
| Meaning | Evenly mixed | Not evenly mixed |
| Appearance | Looks the same everywhere | Looks different in different parts |
| Can you see separate parts? | No | Yes |
| Examples | Salt water, sugar water, air | Sand + water, fruit salad |
| Easy Tip | “Homo” = same | “Hetero” = different |
Quick Tip to Remember:
- Homogeneous = same (everything blends)
- Heterogeneous = different (parts stay separate)
Common Mistakes and How to Avoid Them
❌ Mistake 1: Calling salt water a heterogeneous mixture
Why it happens: People think because salt is solid and water is liquid, the mixture must be uneven.
Correction: Salt dissolves completely → mixture becomes homogeneous.
❌ Mistake 2: Thinking all mixtures where something disappears are homogeneous
Why it happens: Some substances don’t dissolve fully.
Correction: If you can still see particles (like muddy water), it’s heterogeneous.
❌ Mistake 3: Mixing the spelling “heterogeneo”
Many people search for “is salt water homogeneous or heterogeneo”, but the correct spelling is heterogeneous.
Tip: “Hetero” + “geneous” = different kinds
When Do We Use “Homogeneous”?
Use homogeneous when:
- the mixture looks the same from every angle
- you cannot see any particles
- the substance dissolves fully
- the mixture tastes or feels uniform
Examples:
- The scientist prepared a homogeneous salt water solution.
- Air is a homogeneous mixture of gases.
- The juice was so well mixed that it looked completely homogeneous.
- Perfume is a homogeneous liquid mixture.
- The teacher showed us a homogeneous sugar solution during class.
Real-Life Situations:
- Tea after sugar dissolves
- Milk (in most cases, due to processing)
- Soft drinks like Sprite or Coke
When Do We Use “Heterogeneous”?
Use heterogeneous when:
- the parts are clearly different
- the mixture doesn’t blend evenly
- you can see chunks, grains, or layers
Examples:
- A bowl of cereal is a heterogeneous mixture.
- Muddy water is heterogeneous because soil pieces float.
- A salad is always heterogeneous.
- Oil and water stay separate, so they are heterogeneous.
- Rocks in sand create a heterogeneous mixture.
Memory Hack:
Think of hetero as “hey, I can see it!”
If you can see the parts → it’s heterogeneous.
Environmental Factors Can Influence Natural Selection Because They Can Increase or Decrease
So… Is Salt Water Homogeneous or Heterogeneous?
Salt water is homogeneous because the salt dissolves completely, making the mixture uniform.
You cannot see the salt. You cannot separate it by simply looking.
No matter where you taste it from, the saltiness is the same.
👉 Therefore, salt water = homogeneous mixture.
Quick Recap: Homogeneous vs Heterogeneous
- Homogeneous:
- Even mixture
- Looks the same everywhere
- Example: salt water
- Heterogeneous:
- Uneven mixture
- Different parts visible
- Example: sand + water
- Salt water is homogeneous, not heterogeneous
- Use the tip: homo = same, hetero = different
Advanced Tips (Optional but Helpful)
1. Origin of the Words
- “Homo” comes from Greek meaning same.
- “Hetero” comes from Greek meaning different.
2. Use in Exams and Essays
Science questions often ask:
- Identify whether this mixture is homogeneous or heterogeneous.
- Explain why salt water is homogeneous.
3. Use in Real Science
Chemists call homogeneous mixtures solutions.
Salt water = salt solution.
4. Why It Matters
Knowing the difference helps in:
- chemistry experiments
- cooking
- school projects
- understanding everyday materials
Mini Quiz (Test Yourself!)
Fill in the blanks:
- Salt water is a __________ mixture.
- A fruit salad is __________.
- A mixture that looks the same everywhere is called __________.
- Sand in water makes a __________ mixture.
- Air is a __________ mixture of gases.
- When you can see different parts, the mixture is __________.
- Sugar water is always __________.
(Answers: homogeneous, heterogeneous, homogeneous, heterogeneous, homogeneous, heterogeneous, homogeneous)
5 FAQs
1. Why is salt water homogeneous?
Because salt dissolves completely, making the mixture evenly spread.
2. Can salt water ever become heterogeneous?
Yes—if too much salt is added and some crystals remain undissolved.
3. Is sugar water homogeneous or heterogeneous?
Homogeneous, as long as the sugar fully dissolves.
4. What is the easiest way to identify a heterogeneous mixture?
Look for visible parts or layers. If you can see differences, it’s heterogeneous.
5. Is sea water homogeneous?
Mostly homogeneous, but it can be heterogeneous in shallow areas with sand, mud, or floating particles.
Conclusion
Now you clearly understand whether salt water is homogeneous or heterogeneo and why many people get confused. The words sound long, but the idea is simple: homogeneous means same, heterogeneous means different. With real-life examples and easy tips, you can confidently use these terms in school, writing, or science.
Keep practicing with mixtures around you—juice, cereal, soil, or tea. The more you observe, the easier it becomes.

Henry Vale is a tech-savvy content creator at WordContrast.com, known for his in-depth guides and how-to articles. With a background in digital media and a passion for innovation, Henry focuses on simplifying technology for everyday users. His mission is to empower readers with clear, actionable knowledge they can use to improve their digital lives.